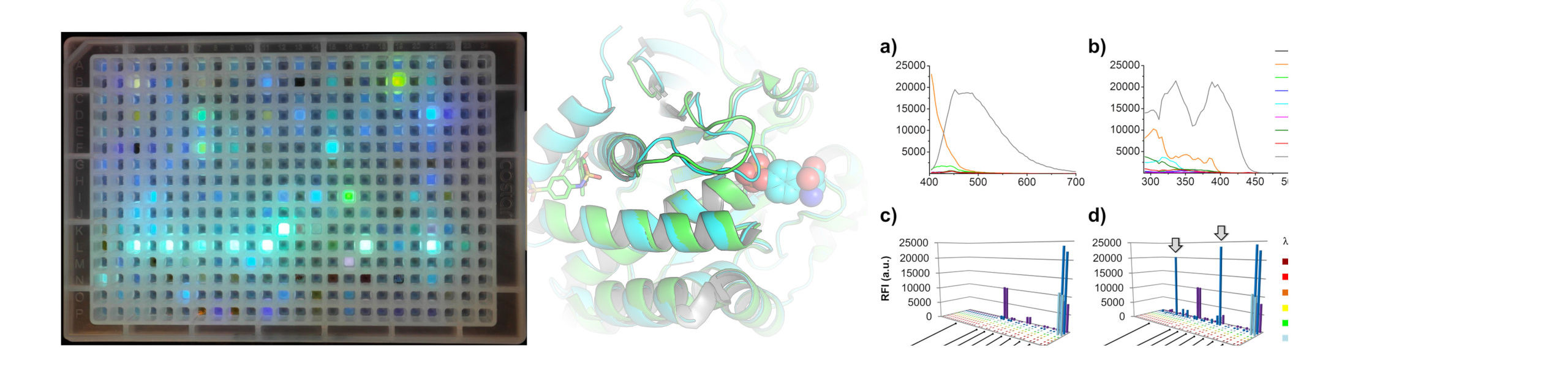
FLiP: Fluorescent Labels in Phosphatase

Normal cellular function, such as signal transduction, is largely controlled by the reversible phosphorylation of cellular proteins catalyzed by two major classes of enzymes, kinases and phosphatases. A misbalance in this complex and dynamic interplay leads to a variety of severe diseases, such as cancer, inflammation, or autoimmune diseases. This makes kinases as well as phosphatases equally attractive targets for therapeutic manipulation by small molecules. While the development of kinase inhibitors has resulted in several blockbuster drugs, such as imatinib, with remarkable success in the clinic and sales of many billions of U.S. dollars per year, not a single phosphatase inhibitor has yet been approved for clinical use. Similar to the kinase world, substrate-competitive phosphatase inhibitors have been developed but were not suitable for further development into clinical candidates due to their charge and limited selectivity. Research efforts, therefore, have shifted to the exploitation of allosteric sites that can regulate phosphatase activity and may enable the discovery of novel modulators of phosphatase activity with much improved pharmacological properties. However, assay systems, which enable the straightforward discovery of these inhibitor types, are missing. Here, we present a novel binding assay capable of detecting ligands of an allosteric pocket of the protein tyrosine phosphatase 1B. This assay is suitable for high-throughput screening and selectively detects ligands which bind to this unique site with a clear discrimination from substrate-competitive ligands.